FIRST CASE REPORT OF PARTIAL RECOVERY OF VISUAL FUNCTION IN A BLIND PATIENT WITH LATE STAGE RETINITIS PIGMENTOSA, USING EVENT-BASED VISION SENSOR
Faster and smoother visual experience
no time spent on re-encoding images
High temporal resolution and low data rate
sparse data generated from bio-inspired sensor
High robustness to lighting conditions
higher dynamic range than conventional sensors
Prophesee’s neuromorphic Event-Based Vision solutions stem from in-depth human vision study. That is why we are particularly excited to see the success of our friends at GenSight Biologics in restoring sight to the blind.
A recent study published in Nature Medicine, in partnership with The University of Basel-UPMC, reported the first case of partial functional recovery in a neurodegenerative disease after optogenetic therapy.
INTRODUCTION TO GENSIGHT
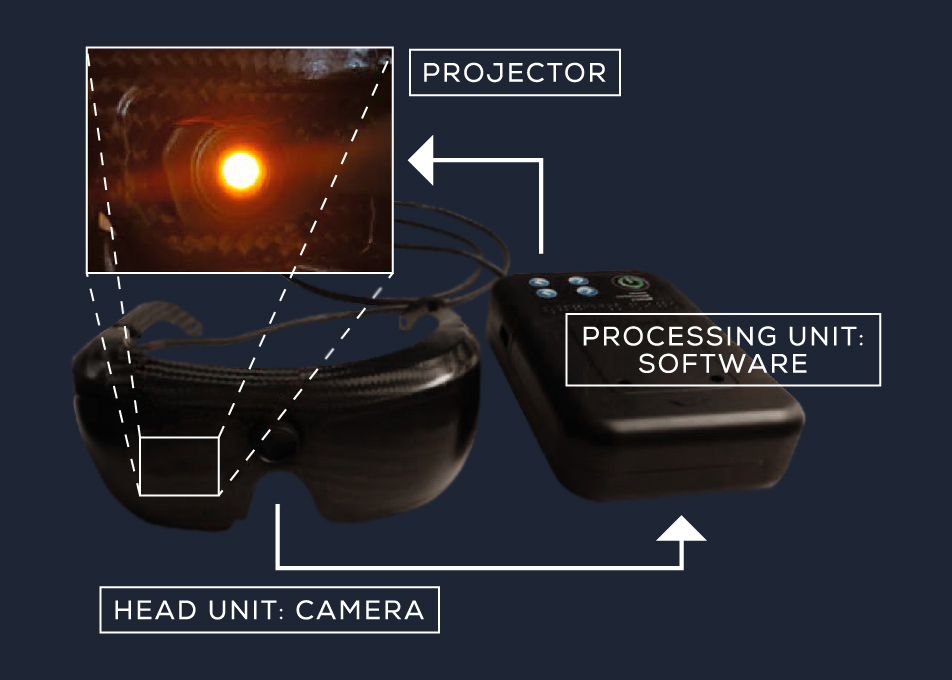
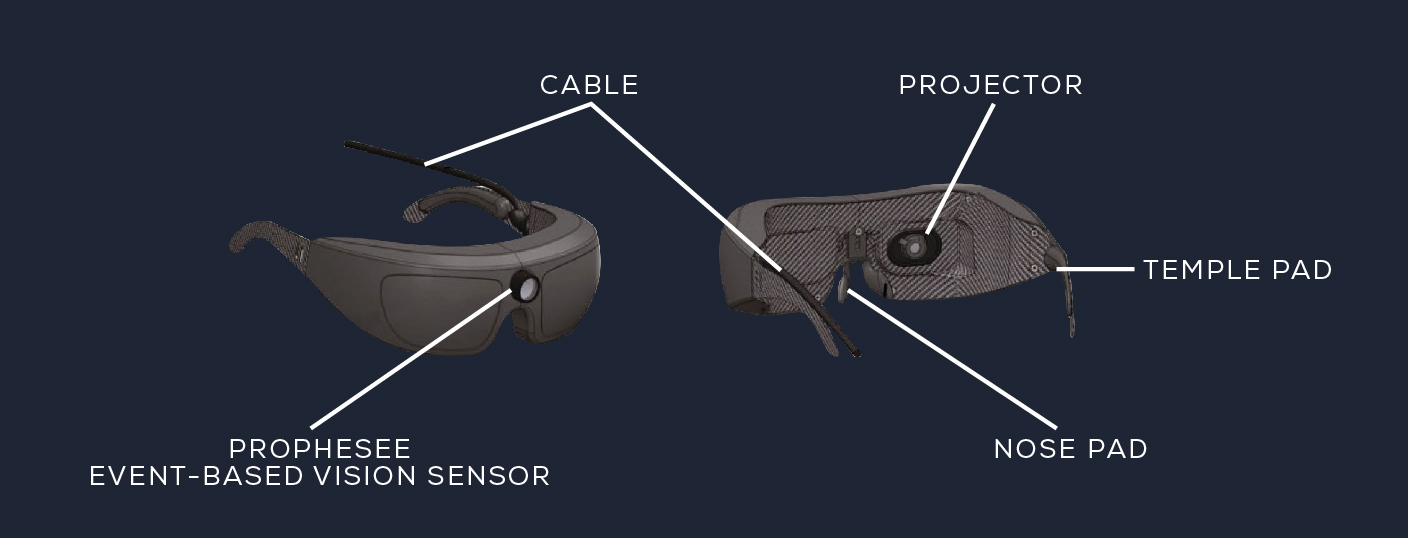
Our Metavision® sensing technologies were used in a light-stimulating medical device (in the form of goggles) and combined with gene therapy to treat a patient with Retinitis Pigmentosa.
A team at The University of Basel-UPMC used optogenetics therapies combined with tech from Gensight to study the treatment of hereditary blindness diseases like Retinitis Pigmentosa, which afflicts 1 in 4,000 people born in the US every year.
OPTOGENETICS
Optogenetics is a biological technique that involves the use of light to control neurons that have been genetically modified to express light-sensitive ion channels. In this study the research team injected a gene isolated from a light-sensing species of green algae into one of the patient’s eyes.
Dr. José-Alain Sahel, Distinguished Professor and Chairman of the Department of Ophthalmology at the University of Pittsburgh School of Medicine, co-founder of Gensight Biologics and co-lead investigator for the PIONEER study explained in an article this was discovered in the late ‘90s and early 2000s.
These proteins exist in algae, capturing light and triggering an electrical response that enables the algae to move towards or retract from light, and it’s a single protein so it’s a very fast response.
A few months after injection, cells are able to detect light and carry the subsequent electrical charge down to the optic nerve. However, they are most sensitive to high intensity light, specifically 590nm wavelength (amber).
To address this issue GenSight developed goggles using our Event-Based Vision sensing technologies to be able to capture this range of light as well as detect changes in light sensitivity. The pixel by pixel function captures only changes, functioning like the human eye.
THE EXPERIMENT
Seven months after training began using the goggles, the patient reported improvements in vision. During tests he was able to identify a notebook (92% of the time) and a pencil case (36% of the time) on a table in front of him. He also correctly counted the number of tumblers on the table (63% of the time). Without the aid of his goggles, the patient successfully completed these tasks zero percent of the time.
Though very preliminary, the current study is a huge step forward for applying this technique and treating patients with neurodegenerative diseases and more. The study was supported by GenSight Biologics; French Programme Investissements d’Avenir IHU FOReSIGHT, Foundation Fighting Blindness and Fédération des Aveugles de France and French National Research Agency.
Change detection in light sensitivity
with event-based vision technology
Correctly located & touched a large and small object
on a table in front of him 92% & 36% of the time respectively
Counted the number of tumblers
placed on the table with 63% accuracy
ABOUT GENSIGHT BIOLOGICS
GenSight Biologics S.A. is a clinical-stage biopharma company focused on developing and commercializing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders. GenSight Biologics’ pipeline leverages two core technology platforms, the Mitochondrial Targeting Sequence (MTS) and optogenetics, to help preserve or restore vision in subjects suffering from blinding retinal diseases.
GenSight Biologics’ lead product candidate, LUMEVOQ® (GS010; lenadogene nolparvovec), has been submitted for marketing approval in Europe for the treatment of Leber Hereditary Optic Neuropathy (LHON), a rare mitochondrial disease affecting primarily teens and young adults that leads to irreversible blindness. Using its gene therapy-based approach, GenSight Biologics’ product candidates are designed to be administered in a single treatment to each eye by intravitreal injection to offer subjects a sustainable functional visual recovery.
ABOUT PROPHESEE INVENTORS COMMUNITY
Since 2014, a network of researchers, start-ups and companies have shown incredible imagination and innovation using Prophesee’s neuromorphic vision technology.
This has created an Event-Based ecosystem of inventors sharing their work and ideas. Their creativity with Prophesee’s technology inspires us.
We are gathering them here to inspire future inventors in turn, in the hope that, like the projects here, they create something new together and reveal the invisible.